|
Our diverse casework spans portfolio prioritization, development pathway and resource optimization, market assessment, product pricing and launch strategies, market segment growth strategies, sales and marketing strategies, franchise development and patent expiration strategies. Most of our studies combine sophisticated quantitative analytics with significant qualitative fact-finding and synthesis to inform strategic decision-making. We serve many of the industry leaders, including the top 20 global biopharmaceutical companies. We pride ourselves on a team-based approach that is client focused and impact oriented. We listen to client needs, help shape hypotheses, analyze data, and advise on the strategy of life-changing therapies, empowering better decisions to create long-term value for patients. We offer an unparalleled depth and breadth of advisory service expertise combined with scientific and technical capabilities across strategic and therapeutic areas. For more than 30 years, we have provided high-impact strategic advice and analytical services, helping our clients solve their most complex challenges across the patient journey and franchise lifecycle to bring preventions and treatments to those that need them. When the combination process is complete, the following Results window appears.Ĭlick OK to complete the process and add the combined study to the main window, as shown below.Putnam, an Inizio Advisory Company, is a premier strategic consulting partner for pharmaceutical and life science companies serving a range of biopharmaceutical, diagnostics, medtech, and private equity/venture capital clients. Specify the desired options and click Run. Note: In cases where there are variables that cannot be combined, this report generates a report listing those variables. Selecting this option enables future snapshot comparisons of study data collected and recorded at different intervals during the study period.Īttempt study combinations when study variables do not matchĬheck this option to combine the selected studies in spite of variable name differences between them. The ADaM Folder must contain at least one domain.įor ADaM BDS Findings data, both PARAMCD and either AVAL or AVALC must be present. The same folder cannot be specified in the SDTM or SEND Folder and ADaM Folder fields.ĭata sets can either be SAS data sets or SAS transport files. The SDTM or SEND Folder must contain at least one domain. 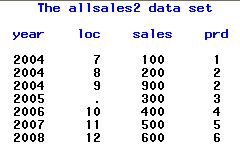
Use the text box to specify the location and name of the folder. Used to combine supplement domains for a study if there are supp domains available. Note: In most cases, the studies are already selected from the Studies tab. Used to select the studies to be combined. Solved: Combine two datasets that have no common variables - SAS Support Communities Solved: Hi, I have a problem that need to combine (or merge) two datesets that have no common variables. The study name must conform to the Rules for Study Names. 
Links to specific documentation for each of the options are provided in the following table: Specify the names of the new study and the SDTM and ADaM folders and click Run. Select the two studies to be combined and click. This option can combine studies with SAS data sets, SAS transport files, or both. The new input data sets are then used to add the combined study to JMP Clinical. The USUBJID values of the new input data sets are unchanged. Running this report preserves all original data from studies to be combined, and creates new input data sets by appending rows from each corresponding parent data set. You can use the following basic syntax to concatenate datasets in SAS: /concatenate two datasets into one/ data data3 set data1 data2 run The following example shows how to use this syntax in practice. 
This report creates a new study by combining two existing studies.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
